
The mass of an electron is 9.1xx10^(-31) kg and velocity is 2.99xx10^(10) cm s^(-1). The wavelenth of the electron will be
The mass of an electron can be expressed as (A) 0.512 MeV (B) 8.19 × 10–14J/c2 (C) 9.1 × 10–31 kg (D) 0.00055 amu

GRE Physics - ✊#Calculation_Using_Concept_E=mc^2 ✋#Question Calculate the kinetic energy and total energy of electron moving with velocity 0.98 times the velocity of light in the laboratory system. #Note_To_Understand. ✌Relativistic mass: Mass increased

31) The mass of an electron is approximately 1/1836 times the mass of A) 1/1H B) 2/1H c) 3/1H D) - Brainly.com

SOLVED: Relative to the mass of the nucleus of an atom, the mass of the electron is A) always much smaller: D) dependent upon the element: about the same B) always much






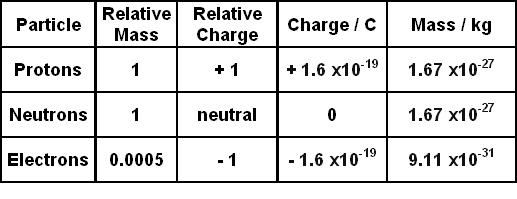









.PNG)