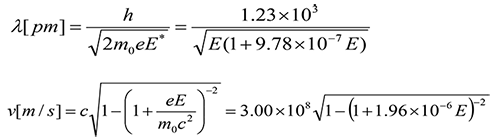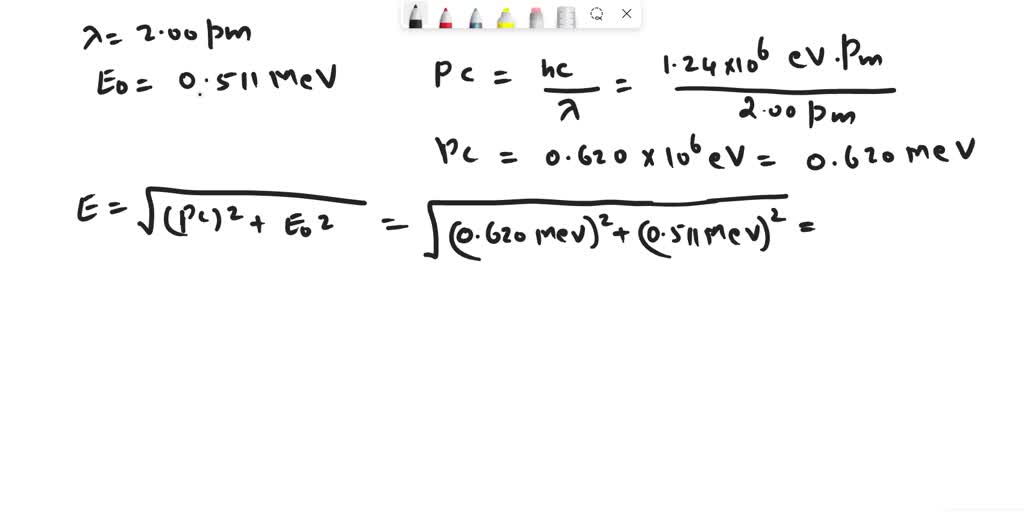
SOLVED: An electron has a de Broglie wavelength of 2.00 pm. Find its kinetic energy and the phase and group velocities of its de-Broglie waves.

Calculate the de Broglie wavelengths of an electron and a proton having same kinetic energy of 100 eV .
Establish formula to find de-Broglie wavelength of electron, proton and α-particle and uncharged particles. - Sarthaks eConnect | Largest Online Education Community

If a proton and an electron have the same de Broglie wavelengths, which one is moving faster? - Quora

explain this properly 15 The de Broglie wavelength of electron of He* ion is 3 329 A If - Chemistry - Structure of Atom - 12893343 | Meritnation.com






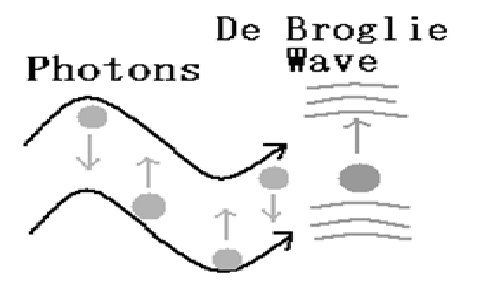




![Solved Problems - Engineering Physics [Book] Solved Problems - Engineering Physics [Book]](https://www.oreilly.com/api/v2/epubs/9788131775073/files/images/co4n066.png)